Save Our Shells – Bubbles Protocol
IV: This is the carbon dioxide that is being blown into the water. It stays constant and doesn’t change.
DV: This is the water. It changes from hot, cold, then room temperature saltwater and also tap water. This is what’s being changed and what we are watching
Ocean acidification occurs when there is too much carbon being absorbed by the ocean. There has been a large increase in the amount of carbon in the atmosphere, which gets absorbed by the ocean. This increase in carbon has been the cause of industrialization which increased the need for fossil fuels. Fossil fuels, such as coal or oil, are whats causing this spike in carbon in our atmosphere.
This increase in carbon causes the water to have a more acidic pH level. This acidity can cause many problems within the ocean ecosystem. One thing is it can cause the levels of phytoplankton to decrease, which are essential to the ecosystem.
In this lab we will be blowing carbon dioxide into different types of water: tap, and hot, cold, and room temperature saltwater. What we are trying to find out is how the carbon dioxide effects the water and also how it effects the different types of water.
Pre-Lab
Questions:
1. We will be blowing carbon dioxide into the water.
2. The water begins to absorb it, making it more acidic.
3. We are measuring change with the universal indicator, which will change colors based on pH levels.
4. Measuring the pH of the water tells us if the water is basic or more acidic.
5. I think it will effect it greatly and cause the saltwater to become more acidic.
I predict that when we blow carbon dioxide into the water, the room temperature water will change quicker than the others.
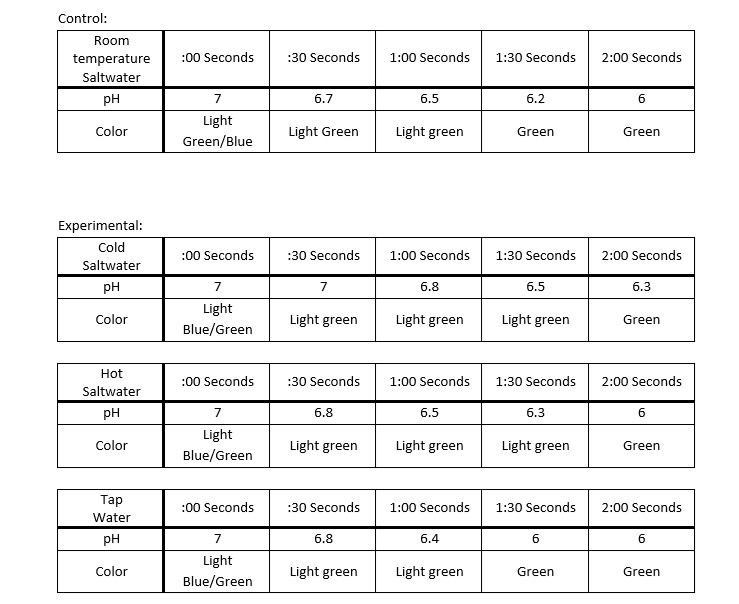
What we will be doing in this lab is having 100 mL beakers of tap water and hot, cold, and room temperature saltwater. In these beakers we will add the universal indicator. We will then insert straws into them and continuously blow in carbon dioxide into them for two minutes. At thirty second intervals, we will identify and record the color and its pH level.
Post-Lab
Questions:
1. Blowing through the straws, you are adding carbon dioxide. It changed the pH level by causing the water to become more acidic.
2. The universal indicator tells us if the water is basic or acidic.
3. It tells us that with a constant flow of carbon, the oceans could quickly become more acidic.
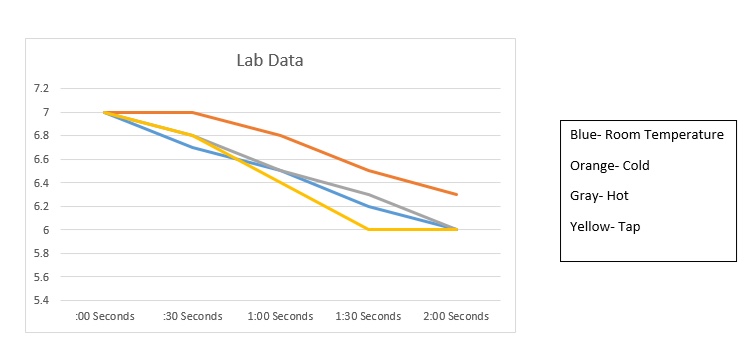
4. Based on my results the salt effects it the most.
My hypothesis was incorrect. I predicted that the room temperature water would change the most, when in fact the tap water is the one that did. Although room temperature was the second one to change the most. This lab showed me how oceans constant contact with carbon dioxide can have a negative effect. How quickly I say the acidic level can change really showed how harmful fossil fuels can be.
IV: This is the carbon dioxide that is being blown into the water. It stays constant and doesn’t change.
DV: This is the water. It changes from hot, cold, then room temperature saltwater and also tap water. This is what’s being changed and what we are watching
Ocean acidification occurs when there is too much carbon being absorbed by the ocean. There has been a large increase in the amount of carbon in the atmosphere, which gets absorbed by the ocean. This increase in carbon has been the cause of industrialization which increased the need for fossil fuels. Fossil fuels, such as coal or oil, are whats causing this spike in carbon in our atmosphere.
This increase in carbon causes the water to have a more acidic pH level. This acidity can cause many problems within the ocean ecosystem. One thing is it can cause the levels of phytoplankton to decrease, which are essential to the ecosystem.
In this lab we will be blowing carbon dioxide into different types of water: tap, and hot, cold, and room temperature saltwater. What we are trying to find out is how the carbon dioxide effects the water and also how it effects the different types of water.
Pre-Lab
Questions:
1. We will be blowing carbon dioxide into the water.
2. The water begins to absorb it, making it more acidic.
3. We are measuring change with the universal indicator, which will change colors based on pH levels.
4. Measuring the pH of the water tells us if the water is basic or more acidic.
5. I think it will effect it greatly and cause the saltwater to become more acidic.
I predict that when we blow carbon dioxide into the water, the room temperature water will change quicker than the others.
What we will be doing in this lab is having 100 mL beakers of tap water and hot, cold, and room temperature saltwater. In these beakers we will add the universal indicator. We will then insert straws into them and continuously blow in carbon dioxide into them for two minutes. At thirty second intervals, we will identify and record the color and its pH level.
Post-Lab
Questions:
1. Blowing through the straws, you are adding carbon dioxide. It changed the pH level by causing the water to become more acidic.
2. The universal indicator tells us if the water is basic or acidic.
3. It tells us that with a constant flow of carbon, the oceans could quickly become more acidic.
4. Based on my results the salt effects it the most.
My hypothesis was incorrect. I predicted that the room temperature water would change the most, when in fact the tap water is the one that did. Although room temperature was the second one to change the most. This lab showed me how oceans constant contact with carbon dioxide can have a negative effect. How quickly I say the acidic level can change really showed how harmful fossil fuels can be.
Shells Protocol
Pre-Lab Questions:
1. Organisms create their shells using calcium carbonate.
2. I expect the shells to become weak and fragile because of the acidity.
3. The sources of carbon dioxide is fossil fuels, cars, and industrialization. Fossil fuels is the leading contributor to the rise in
acidity.
My hypothesis is that the shell that has had a higher concentration of acidity will break the easiest. I believe this because if shells have a long exposer to acidity, their shells won’t fully develop making the shell weaker.
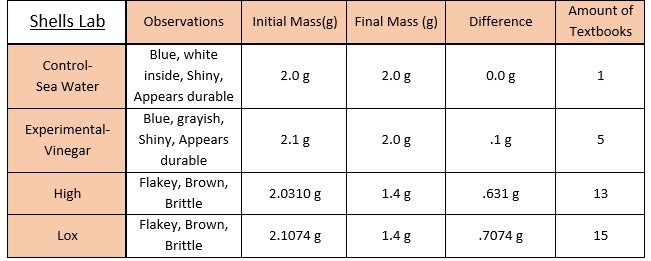
What we did in this lab is have two beakers with 150mL of one with vinegar and one with salt water. We were then given four shells: two untreated, one treated with a higher/longer time in acidity, and one treated with a lower/shorter time in acidity. We then labeled two of the untreated shells with one having an E and the other having a C. The E shell (experimental) was put into the vinegar for 30 minutes and the C shell (control) was put in the saltwater for 30 minutes also. Once they were done being in the water for thirty minutes, we took the four shells and tested their strength. We did this by seeing how many textbook each different shell could hold.
Post-Lab Questions:
1. We knew a reaction was happening with the sell in the vinegar because you immediately could see it being covered in tiny bubbles.
2. It relates to animals because possibly it made the shell weaker, and that’s what is does to animals. Also maybe because you could see the effects it has as you can see with the animals.
3. Shelled organisms shells would begin to weaken, just as the shell in the vinegar had done.
4. The primary function of the shell for these animals is protection and if their shell becomes weak their protection is weak, allowing them to easily be eaten, killed,
or anything else.
5. Yes, it does cost the animal energy to rebuild their shell. By using more energy, they are in more need of oxygen. The use of too much oxygen can cause problems too.
Conclusion:
Yes, my initial hypothesis was correct. I predicted that the shell with the higher concentration with beak the easiest, which was proved to be true. Between the high and low, the high shell had the most exposer and it broke first. Between the Control and Experimental, the Experimental had the most exposer to acidity and it broke first also.
Pre-Lab Questions:
1. Organisms create their shells using calcium carbonate.
2. I expect the shells to become weak and fragile because of the acidity.
3. The sources of carbon dioxide is fossil fuels, cars, and industrialization. Fossil fuels is the leading contributor to the rise in
acidity.
My hypothesis is that the shell that has had a higher concentration of acidity will break the easiest. I believe this because if shells have a long exposer to acidity, their shells won’t fully develop making the shell weaker.
What we did in this lab is have two beakers with 150mL of one with vinegar and one with salt water. We were then given four shells: two untreated, one treated with a higher/longer time in acidity, and one treated with a lower/shorter time in acidity. We then labeled two of the untreated shells with one having an E and the other having a C. The E shell (experimental) was put into the vinegar for 30 minutes and the C shell (control) was put in the saltwater for 30 minutes also. Once they were done being in the water for thirty minutes, we took the four shells and tested their strength. We did this by seeing how many textbook each different shell could hold.
Post-Lab Questions:
1. We knew a reaction was happening with the sell in the vinegar because you immediately could see it being covered in tiny bubbles.
2. It relates to animals because possibly it made the shell weaker, and that’s what is does to animals. Also maybe because you could see the effects it has as you can see with the animals.
3. Shelled organisms shells would begin to weaken, just as the shell in the vinegar had done.
4. The primary function of the shell for these animals is protection and if their shell becomes weak their protection is weak, allowing them to easily be eaten, killed,
or anything else.
5. Yes, it does cost the animal energy to rebuild their shell. By using more energy, they are in more need of oxygen. The use of too much oxygen can cause problems too.
Conclusion:
Yes, my initial hypothesis was correct. I predicted that the shell with the higher concentration with beak the easiest, which was proved to be true. Between the high and low, the high shell had the most exposer and it broke first. Between the Control and Experimental, the Experimental had the most exposer to acidity and it broke first also.